Secretion and the Extracellular Matrix
Reading
Secretory Pathway
The importance of the cell membrane to the viability and function of cells raises a couple of important questions. First, how do the protein channels, receptors, and adhesion proteins get into the membrane when proteins cannot diffuse across a membrane? Second, how do cells replace those phospholipids and proteins in a cell membrane that are damaged and have loss part or all of their function?
The secretory pathway allow cells to deliver phospholipids and proteins to the cell membrane and some organelles (e.g., lysosome, secretory granules). The secretory pathway comprises a set of distinct membrane-bound organelles that synthesize proteins and phospholipids, modify them, and delivery them to their final destination.
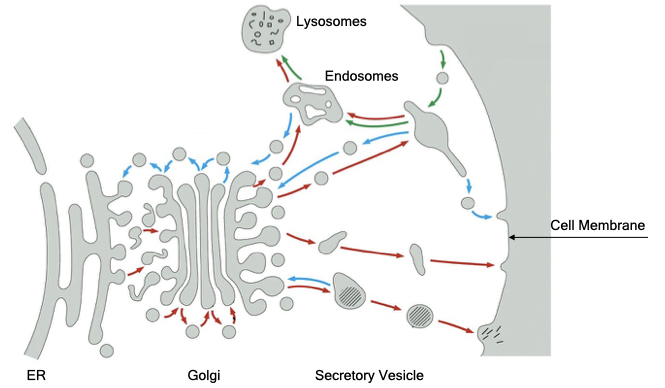
The diagram below provides an overview of the membrane-bound organelles in the secretory pathway. The entry point for the secretory pathway is the endoplasmic reticulum. Here, proteins are synthesized and inserted into or across the phospholipid bilayer. In addition, most steps in the synthesis of phospholipids occurs in the endoplasmic reticulum. From the endoplasmic reticulum, proteins and phospholipids are delivered to the Golgi.
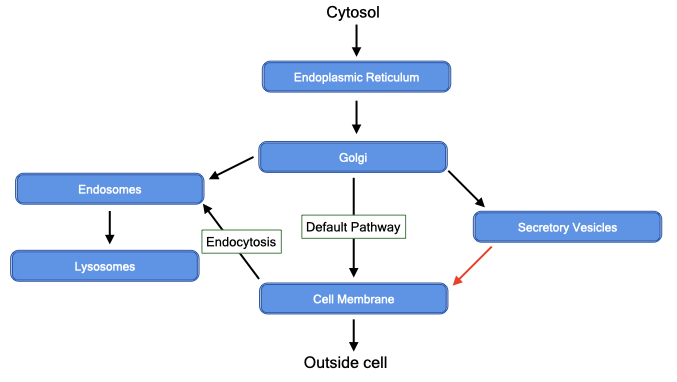
The Golgi performs two essential functions. First, it modifies sugar side chains on proteins that were added in the endoplasmic reticulum. Second, it sorts proteins to their final destination, often by recognizing signal sequences, which are short stretches of amino acids within proteins. By default, proteins and lipids are delivered to the cell membrane but can be diverted to internal organelles, such as lysosomes or secretory vesicles. Secretory vesicles store proteins and other molecules within a membrane organelle in the cytoplasm of cells. When stimulated, the vesicles fuse with the cell membrane to release their contents to the external environment.
In addition to delivering material to the cell membrane, cells can internalize portions of their cell membranes through a process called endocytosis. Endocytosis allows cells to remove specific proteins from the cell membranes or take up soluble material from the external environment.
Protein Import into the Endoplasmic Reticulum
The first step in the secretory pathway is the entry of proteins into the endoplasmic reticulum (ER). Protein import into the ER requires coordination between the translation of protein from mRNA and translocation of that protein across the membrane the ER.
All proteins are synthesized from mRNA by ribosomes in the cytosol (fluid portion of cytoplasm) of cells. Proteins destined for the secretory pathway contain a signal sequence at their N-terminus. When this signal sequence emerges from the ribosome, a complex of 6 proteins and one RNA, called the signal-recognition particle (SRP), binds to the signal sequence. After binding to the signal sequence, SRP pauses translation of the protein.
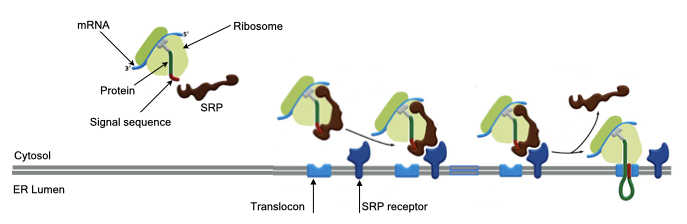
The ribosome with SRP is next bound to the surface of the ER membrane through the interaction between SRP and a receptor embedded in the ER membrane called SRP receptor. The protein being synthesize by the ribosome cannot diffuse across the phospolipid bilayer of the ER membrane, so SRP receptor resides next to a protein channel called the translocon. The translocon forms an aqueous pore across the ER membrane through which a protein can pass from the cytosol into the ER.
The interaction between SRP and its receptor causes SRP to release from the nascent protein and ribosome. This allows translation to continue and the energy of translation pushes the protein through the translocon into the ER.
Soluble and Integral Membrane Proteins
Synthesis and translocation of proteins across the ER membrane can generate both soluble proteins that reside entirely within the lumen of the ER and integral membrane proteins that span the ER membrane at least once and up to several times. Usually, soluble proteins are secreted into the external environment, whereas integral membrane proteins that reach the cell membrane make up the receptors, channels, pumps and adhesion proteins that generate specific functions for the cell.
Soluble proteins contain one signal sequence that initiates translocation of the protein entirely across the ER membrane. After translocation, the bulk of the protein is cleaved from the signal sequence which releases the protein into the lumen of the ER.
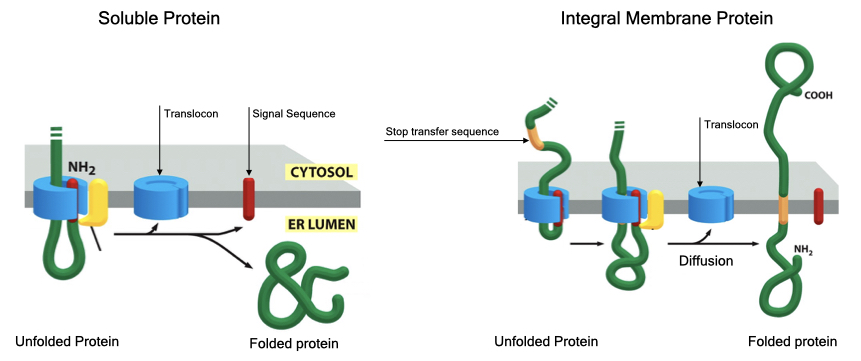
Integral membrane proteins also contain a signal sequence that mediates translocation but contain an additional signal sequence called a stop-transfer sequence. The stop transfer sequence pauses translocation through the translocon and allows the protein to diffuses out of the translocon with the stop transfer sequence spanning the lipid bilayer. The signal sequence is then cleaved from the protein.
Proteins that span the membrane more than once contain a start transfer sequence downstream of the stop-transfer sequence that reinitiates translocation. A second stop-transfer sequence stops translocation and forms the second membrane-spanning region. These alternating start and stop transfer sequences can be repeated several times to generate proteins that span the ER membrane several times.
Protein Folding
The pore in the translocon can only accommodate proteins in an unfolded state. Proteins moving through the translocon must fold into a proper structure before they are allowed to leave the ER (the ER has mechanisms to prevent unfolded protein from exiting). Mutations in proteins that inhibit their folding or high levels of synthesis of secreted protein can accumulate unfolded protein in the ER, which can lead to cell death. You will encounter several diseases where one of the causes is due to accumulation of unfolded protein.
Protein Glycosylation
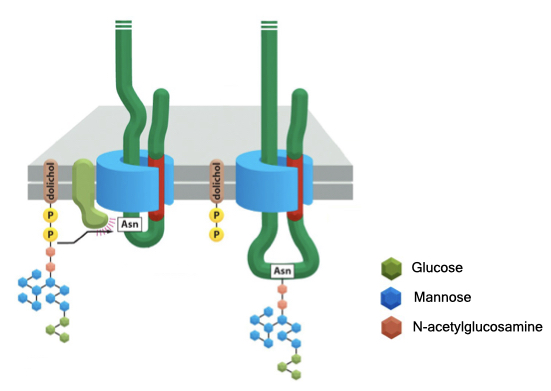
Many proteins that enter the ER are modified by the transfer of a block of sugar residues onto an asparagine residue in the protein. The block of sugars is covalently attached to a nitrogen atom in asparagine and is therefore called N-linked glycosylation. The block of sugars contains a mix of glucose, mannose and N-acetylglucosamine that are assembled into a tree-like structure. The block of sugars serve several functions including protein localization (see below), protein folding and cell adhesion.
Vesicular Transport
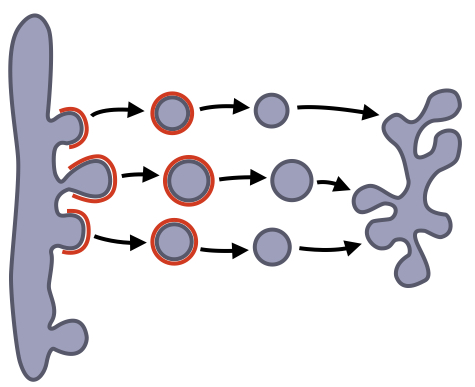
After proteins are imported into the ER, they must be delivered to their appropriate location (e.g. cell membrane, lysosome, etc.) via the secretory pathway. Trafficking of proteins between organelles is mediated by small, membrane-bound vesicles that form from one organelle and then fuse with another organelle.
Vesicular transport between organelles consists of three steps. First, vesicles bud from one organelle (e.g ER). The vesicle is then targeted to the appropriate organelle (e.g Golgi). Finally, the vesicle fuses with the target organelle to mix its contents with the contents of the target organelle. The formation of a vesicle also includes a mechanism for cargo selection because only a subset of proteins in an organelle should leave that organelle to move to another organelle. Some proteins are considered resident proteins because they perform essential functions in an organelle. The formation of vesicles and cargo selection are mediated by proteins that form a coat around the vesicle. After formation of the vesicle, the coat falls off allowing the vesicle to fuse with its target organelle.
Vesicle Targeting and Fusion
Once vesicles form from one organelle, they need to fuse with another organelle or the cell membrane to deliver their cargo. There are many different membrane-bound organelles in the cell, so vesicles need a mechanism to recognize the correct target organelle.
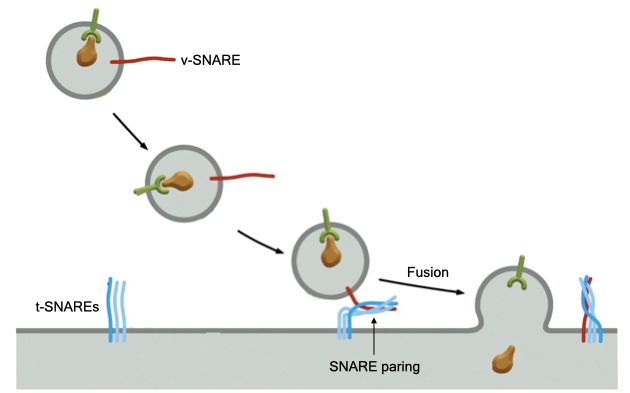
A large family of proteins called SNAREs mediates fusion between vesicles and organelle. SNAREs are transmembrane proteins that reside in both organelles and vesicles. SNAREs in vesicles are called v-SNAREs and SNAREs in organelles are called t-SNAREs (for target). There are over 35 different SNARE proteins allowing the cells to mark different types of organelles and vesicles with a unique set of SNAREs. Certain v-SNAREs and t-SNAREs interact or pair to generate a specific interaction between a vesicle with its target organelle. The energy of pairing between SNAREs is thought to drive the fusion of the vesicle membrane and organelle membrane. Fusion mixes the contents of the vesicle with the target organelle.
Protein Sorting in the Secretory Pathway
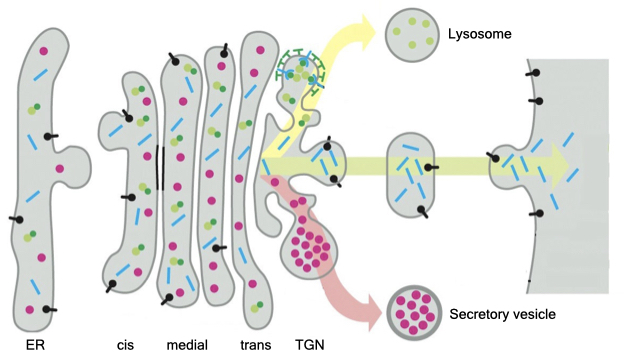
By default, proteins that enter the secretory pathway are delivered to the cell membrane. If the protein is an integral membrane protein, it will reside in the cell membrane (e.g., receptor, channel). If the protein is soluble (does not span the membrane), it will be released to the surrounding environment.
Proteins that need to be delivered to another location (e.g., intracellular organelle) are sorted in the Golgi. The Golgi comprises a series of disc-shaped, membrane-bound organelles called cisternae. Proteins from the ER are delivered to the cis-side and proteins leave the Golgi from the trans-side. If a protein contains a certain signal sequence, the Golgi will divert the protein from delivery to the cell membrane and target the protein to its correct destination.
Protein Trafficking to Lysosomes
Let’s look at important example of how proteins are targeted to an intercellular organelle, the lysosome. Lysosomes are membrane-bound organelles that digest cellular and foreign material. The lumen of the lysosome contains many protein enzymes that breakdown different macromolecules. These enzymes are active at low pH, and proton pumps in the membrane of lysosomes acidifies its lumen. The advantage of using enzymes that are active at low pH is it keeps those enzymes inactive until they reach the lysosome. To perform its myriad of activities the lysosome requires the secretory pathway to deliver a constant supply of several different digestive enzymes. If lysosomes lack one or more of these enzymes, then biological material accumulates in the lysosome, and the lysosome enlarges.
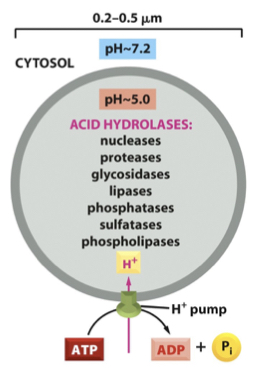
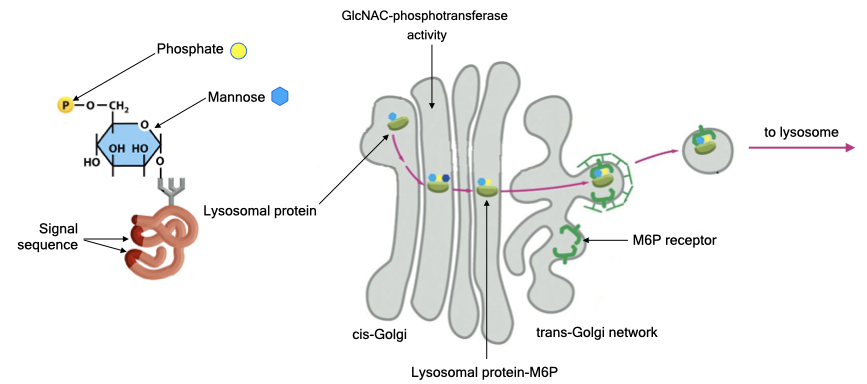
Many enzymes are targeted to the lysosome via a unique sugar modification called mannose 6-phosphate. Recall that proteins are glycosylated in the ER, and mannose 6-phosphate will be added to proteins that are meant for the lysosome.
Proteins destined for the lysosome contain a signal sequence that is recognized by an enzyme in the Golgi called GlcNAC-phosphotransferase. The enzyme catalyzes the transfer of a sugar called N-acetylglucosamine with a phosphate onto the mannose residue of the sugar side chain of the lysosomal protein. The N-acetylglucoamine is eventually removed, leaving mannose-phosphate on the lysosomal protein.
A receptor in the TGN recognizes proteins with mannose-6-phosphate and recruits those proteins into vesicles that will be delivered to lysosomes.
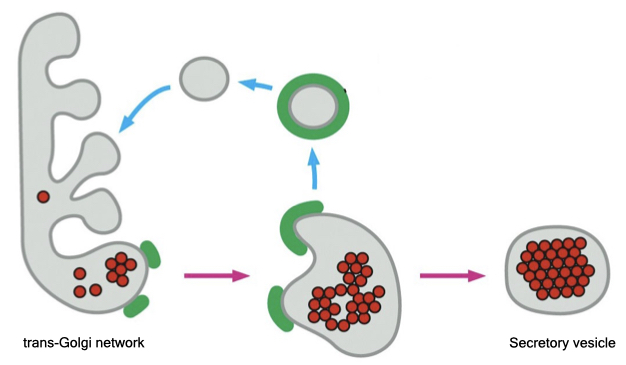
Secretory vesicles
Some cells release proteins and peptides to the external environment and store those components in intracellular vesicles called secretory vesicles. This allows cells to accumulate a large amount of material and quickly release that material upon receiving a signal to do so.
The mechanism of targeting protein to secretory vesicles is less clear than how proteins are trafficked to lysosomes. The process appears to involve a signal patch on proteins that induces them to cluster in the TGN. The aggregated protein is selected by an unknown mechanism into vesicles that bud from the TGN. After budding from the TGN, secretory vesicle undergo a maturation process during which the content of the secretory vesicle is concentrated. Secretory granules do not immediately fuse with the cell membrane but remain in the cytoplasm until an appropriate signal to fuse is received by the cell.
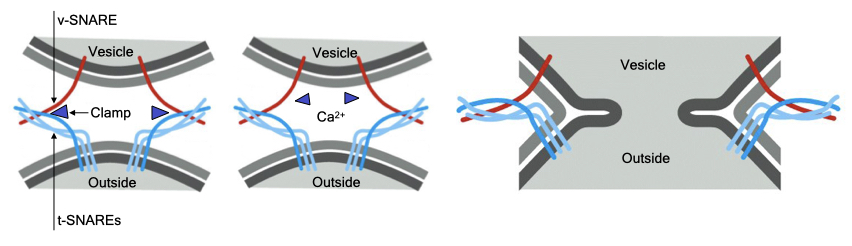
Some secretory vesicles dock at the cell membrane in an arrested state of fusion. Fusion with the cell membrane is completed only when the cell receives an appropriate signal. This is an example of regulated fusion that is common in a variety of different cells.
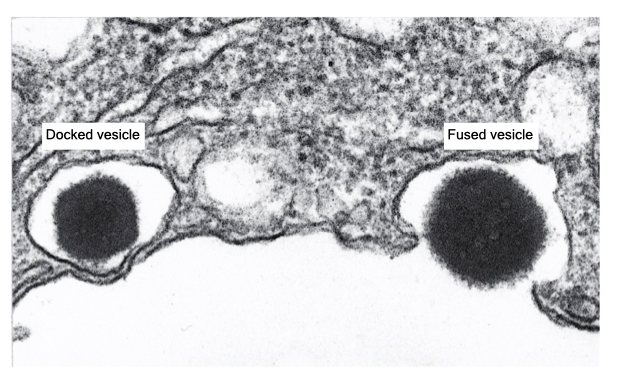
The mechanism of one type of regulated fusion is mediated by the SNARE proteins. The v-SNARE in the granule and t-SNAREs in the cell membrane interact but are prevented from completely pairing by an inhibitory protein called a clamp. When a signal is received, there is an increase in cytosolic calcium that dissociates the clamp from the SNAREs. Now the SNAREs can complete their pairing to drive fusion of the vesicle with the cell membrane. Docking of the granule at the cell membrane allows for rapid release of the vesicle contents in response to stimuli and is the mechanism by which neurons release neurotransmitter.
Endocytosis
Endocytosis comprises several pathways through which cells recycle proteins and lipids in the cell membrane and take up material from external environment. These pathways include pinocytosis, receptor-mediated endocytosis, and phagocytosis.
Pinocytosis
Pinocytosis is process in which cells pinch off a small portion of their cell membranes to form a vesicle. Through pinocytosis the cell removes 1 to 3% of plasma membrane per minute. Pinocytosis is critical for a cell’s viability because over time proteins and lipids in the cell membrane are modified by oxidation or damaged by other factors. If this material was not removed, the function and structural integrity of the cell membrane would decline and potentially cause cell death. Cells use the secretory pathway to replace protein and lipid removed through pinocytosis. Pinocytosis appears to occur randomly along the surface of the cell membrane, so eventually most proteins and lipids are recycled.
Formation of a vesicle in pinocytosis is usually driven by a set of proteins called clathrin. Clathrin proteins form a basket-like coat on the cytoplasmic side of the cell membrane and facilitate formation and pinching off of a vesicle. Once formed, the vesicles are called clathrin-coated vesicles. Eventually, the clathrin coat falls off the vesicle, and the vesicle fuses with the lysosome where its digestive enzymes breakdown the protein and lipids in the vesicle.
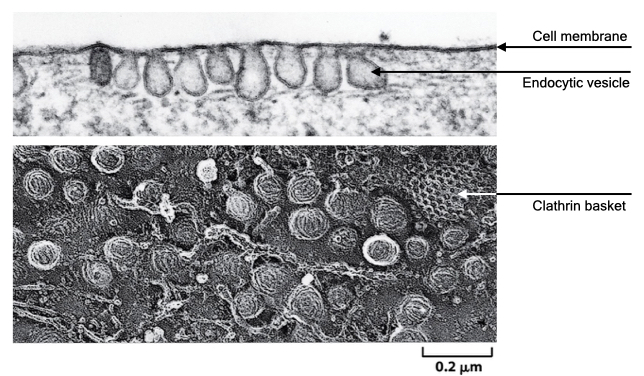
During pinocytosis, cells also take up small amounts of external fluid with the cell membrane, which allows them to sample the external environment.
Receptor-Mediated Endocytosis
Most cells also have a mechanism to remove specific proteins from their cell membranes. Here, clathrin forms at specific sites on the cell membrane and recruits certain proteins into the endocytic vesicles. Cells mark proteins for endocytosis by modifying a domain that faces the cytoplasm. This modification is recognized by the clathrin machinery which form a clathrin coat near the location of modified proteins and recruit other modified proteins into the site. The endocytic vesicle that forms has a high concentration of modified protein.
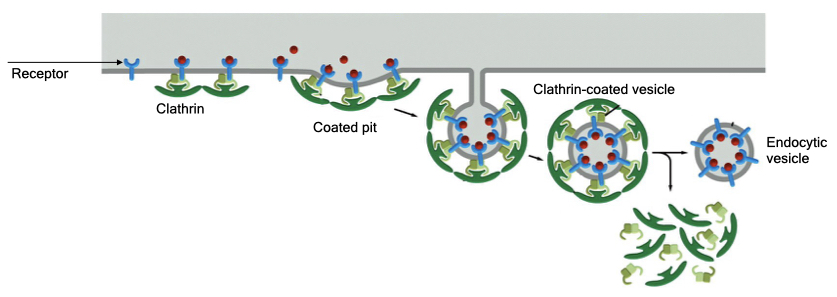
Receptor-mediated endocytosis serves several functions. One is to take up nutrients that cells need to survive. Some receptors bind material that contain essential nutrients. Receptor-mediated endocytosis brings that material into the cell where it can be processed in the lysosome to release the nutrients. Receptor-mediated endocytosis also allows cells to regulate the amount of a specific protein in its cell membrane. This will be important when we discuss cell communication and how cells limit their response to a signal.
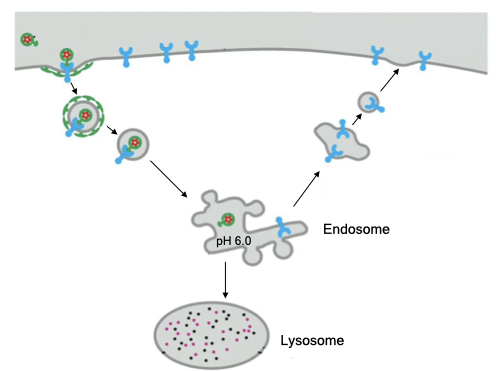
Similar to vesicles that form through pinocytosis, vesicles that form via receptor-mediated endocytosis are targeted to the lysosome. However, the cell also has the option of recycling receptors back to the cell membranes before the vesicle fuses with the lysosome. After the clathrin coat falls off the vesicle, the endocytic vesicle acidifies to dissociate the receptor from its cargo. At this point the vesicle is called an endosome. The receptor can be recycled from the endosome to the cell membrane and the cargo delivered to the lysosome.
Phagocytosis
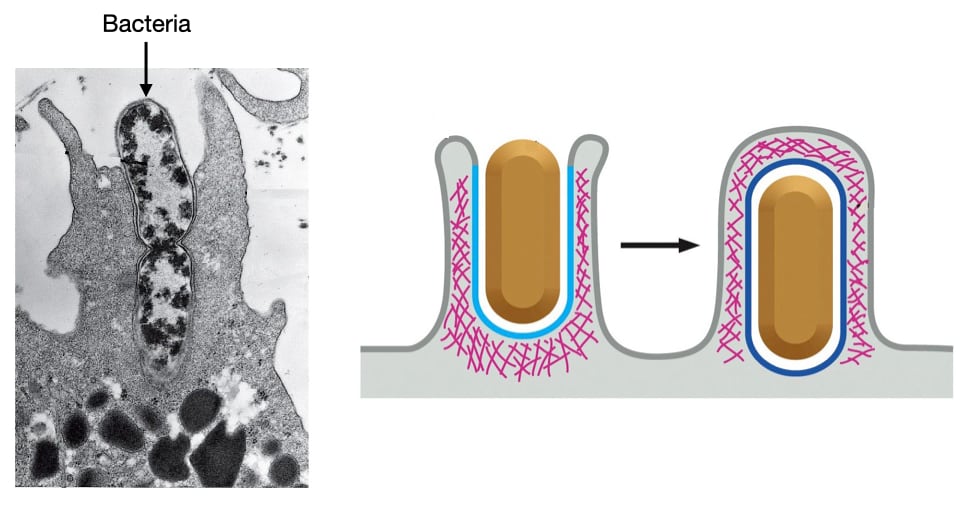
Phagocytosis is a process through which certain cells engulf large objects, such as bacteria or cellular debris. Phagocytosis is usually found only in certain immune cells like macrophages and neutrophils. Phagocytosis is triggered when a macrophage or neutrophil binds a foreign object on its cell membrane. The cell then pushes its cell membrane around object through an actin-dependent process to engulf the object. Once completely surrounded by cell membrane, the object will usually be delivered to the lysosome for degradation.
Extra Cellular Matrix
Functions of Extracellular Matrix
The extracellular matrix (ECM) is a collection of proteins, carbohydrates and fluid that perform a variety of functions in tissues. First, the ECM provides structural support to cells by allowing cells to attach to a common substrate. By connecting to the ECM, cells become integrated into a structural unit that has more robust mechanical properties than if cells were only attached to each other.

In this image nuclei are stained in blue and the ECM is stained in green. Note how the ECM forms a network that surrounds the cells to provide structural support.

ECM also regulates activities within cells in two different ways. First, the mechanical properties of the ECM affect the behavior of cells. An ECM that is stiffer or more resistant to tension elicits different responses in cells compared to an ECM that is more easily deformed. For example, a stiff ECM can induce morphological changes in cells, trigger cell division or differentiation, and alter gene expression.

Cells sense the stiffness of the ECM through contacts they make with components of the ECM. These contacts often aggregate in structures called focal adhesions. Focal adhesions associate with actin filaments within the cytosol of cells.

Cells can generate tension on focal adhesions through actin and myosin filaments. Bipolar myosin filaments crosslink actin filaments that are attached to different focal adhesions. Focal adhesions are linked to the ECM via a set of integral membrane proteins in the cell membrane called integrins, which we’ll discuss in detail later. When the myosin filaments are active, they pull on the actin filaments which in turn pull on the focal adhesions. If the focal adhesions are attached to an ECM that is stiff, the actin and myosin filaments can generate more tension on the focal adhesion because the stiffness of the ECM will resist movement of the focal adhesions. The higher tension on the focal adhesions initiates a signaling pathway which changes cell behavior. If focal adhesions are attached to a less stiff ECM, then the cell will not be able to generate as much internal tension and the signaling pathways will not be activated.

A second way that ECM controls cell behavior is by controlling the availability of signaling molecules. Hormones and other signaling molecules must diffuse through ECM to reach receptors on the surfaces of cells. Some of the components of the ECM bind to signaling molecules which slows their diffusion and in some cases, prevents them from binding to receptors on the surfaces of cells. Thus, ECM can regulate the concentration of signaling molecules that are available to cells and determine how tissues respond to signaling molecules.

Components
There are three classes of protein components in extracellular matrix that largely determine its mechanical properties: collagen fibers, elastic fibers and glycoproteins. In general, these molecules either resist tensile and stretching forces or compressing forces. Collagen is the main component that resist tension. Elastic fibers also resist tension but behaves similar to rubber in that it can be stretched and will recoil after the force is removed. On the other side are glycosaminoglycans that resist compressive forces.

Collagen
Collagen is the most abundant class of proteins and pound for pound some are as strong as steel. There are several different types of collagens and their locations within the body varies. Most collagens, about 80-90% of total collagen, form fibers that provide the most mechanical strength. Aggregation and lateral interactions between the individual fibers increase the mechanical strength.
There are over twenty different types of collagen but the most abundant and medically significant are types I - IV. Types I - III form fibers which gives them greater mechanical strength and ability to resist tension. We’ll discuss these collagens and their tissue location in more detail when we cover connective tissue. Type IV forms a branched network which allows it to form a sheet-like structure. We’ll discuss type IV collagen in more detail when we cover epithelia.

Type I - IV collagens are composed of three polypeptide chains. Each of these polypeptides can be over 1000 amino acids giving the trimer an overall length of 300 nm and a width of 1.5 nm. A collagen trimer can be composed of polypeptides encoded by different genes (Type I and IV) or polypeptides from the same gene (Type II and III). The polypeptides form alpha-helices that wrap around each other to form coiled coil interactions. The sequence for most of the length of a collagen polypeptide is a repeat of 3 amino acids: glycine and usually proline and lysine. Glycine which is the smallest amino acid allows for tight packing of the polypeptides in the trimer. Note the extensive lateral interactions that give the structure its mechanical strength.

Each type of collagen is encoded by one or more genes:
- Type I Collagen: COL1A1 and COL1A2
- Type II Collagen: COL2A1
- Type III Collagen: COL3A1
- Type IV Collagen: COL4A1 - COL4A6
Thus, type I collagen trimers are composed of a mix of A1 and A2 protein.
Many different types of cells can synthesize collagen but one of the most prominent in extracellular matrix is the fibroblast shown here in an electron micrograph. We’l encounter fibroblasts again when we discuss specific tissues and organs.

The synthesis of collagen polypeptides in fibroblasts involves several key steps that allow them to assembly into collagen fibrils. Because collagen is a secreted protein, it is synthesized on ER-bound ribosomes and translocated across the ER membrane during translation. One important feature of collagen is that it contains extra sequence at its N and C-termini called prodomains. These prevent collagen trimers from assembling into fibrils inside the cell which would be catastrophic for the cell.
Collagen undergoes two important modifications in the ER. First, certain prolines and lysines are hydroxylated. These modifications will allow for assembly into trimers and covalent crosslinks between collagen trimers outside the cell. Second, disulfide bonds between collagen polypeptides mediates their assembly into trimers by facilitating interaction between correct collagen proteins.

Once secreted, the prodomains are removed from the trimer by proteases that reside outside fibroblasts to produce the mature collagen trimer. Collagen trimers then self-assemble into fibrils through an entropy-driven process (does not require input of energy).

The interactions between collagen trimers allow them to assemble into fibrils but the interactions are insufficient to account for collagen’s ability to resist tension. Covalent crosslinks between adjacent trimers generates a stronger interaction between trimers. Lysyl oxidase catalyzes a reaction that generates covalent bonds between hydroxlysines in adjacent trimers. Mutations that affect hydroxylation of lysines weakens collagen.

Some collagen fibrils (type I) then aggregate into large bundles called fibers. Note in the image below collagen fibers are shown in cross-section (green circle) and longitudinally (green arrow). In cross-section each dot is a collagen fibril and the collection of fibrils composes a fiber.

This gives type I collagen three levels of lateral interactions:
- Trimer - assembles inside fibroblasts.
- Fibrils - aggregation of trimers outside of fibroblasts and crosslinked by lysyl oxidase..
- Fibers - aggregation of fibrils.
The extensive interactions are in part what make type I so effective at resisting tension.
The proper assembly of collagen is dependent upon two enzymes that act immediately after collagen is synthesized in the ER. Proline and lysyl hydroxylases convert proline and lysine into hydroxylated versions. Hydroxylated proline facilitates packing of individual collagen polypeptides into a trimer and hydroxylated lysine is required to generate covalent attachments between trimers.. Both enzymes require vitamin C as a cofactor and people who don’t consume enough vitamin C will produce collagen that lacks hydroxylated lysines and prolines. Because these trimers cannot be crosslinked, the collagen fibrils will be weaker leading to tissues that are more prone to damage.

Elastic Fibers
Next, we will examine elastic fibers that are often found enmeshed with collagen fibers as shown in this cross section of an artery. The elastic fibers stain dark blue and appear as a wavy line whereas the collagen stains light blue. Elastic fibers have different mechanical properties from collagen. They allow for stretching of tissues under external force, but generate a recoil force when the external force is removed. Elastic fibers are prominent in the walls of arteries especially the aorta. The elastic fibers stretch to allow the aorta to accommodate a large volume of blood during systole. When the pressure drops during diastole, the fibers recoil pushing blood into the circulatory system. Because of elastic fibers a consistent blood pressure is generated in the circulatory system, even though the pumping of the heart delivers blood in a pulsatile fashion.

Elastic fibers are a composite material composed of two primary components: elastin and fibrillin. Both are synthesized by fibroblasts and other cells and are secreted into the surrounding tissue where they assemble into elastic fibers. Fibrillin fibers are thin and arranged in more or less parallel arrays. They are required for correct assembly of elastic fibers. Elastin appears as an amorphous substance. Elastin is what gives elastic fibers its characteristic mechanical properties of stretching and recoiling.

Elastin is the main structural component of elastic fibers. Compared to collagen, elastin is largely unstructured which gives it that amorphous appearance in electron micrographs. Elastin has a hydrophobic domain which mostly lacks structure and an alpha-helical domain. Lysyl oxidase generates covalent attachments between lysines in the alpha-helical domain of adjacent elastin polypeptides. This generates a network of interconnected elastin proteins.

The unstructured nature of elastin accounts for its ability to stretch and recoil. Relaxed fibers largely unstructured and disordered. The hydrophobic domains cluster to avoid water. Tension stretches elastin generating order in hydrophobic domains. When tension is removed, elastin returns to its lower energy state of disordered fibers, causing the elastin network to recoil. The relaxation of elastic fibers does not require input of energy but is due to the transition from a high energy state to a low energy state.

.
Glycosaminoglycans
Extracellular matrix not only resist tensile or pulling forces but also resist compression. The primary component in ECM that responds to compression are a family of molecules called glycosaminoglycans (GAGs). GAGs resist compression by occupying a large volume and retaining water within that space. This is similar to how a plastic bottle filled with water resists compression. An air-filled bottle collapses under applied force because the force expels the air from the bottle. In contrast, a bottle filled with water that is sealed so that it retains water, resists compression from an outside force.

Glycosaminoglycans (GAGs) are the main component that retains water to resist compression. The base component of GAGs is a disaccharide of two different sugars. These disaccharides are joined into polymers that can contain thousands of disaccharides. The sugars that compose the disaccharides differ between GAGs, but a feature they share is a negative charge on one of the sugars. Thus, GAGs are long, negatively-charged polymers. The negative strong charge in GAGs attracts sodium which through osmosis attracts and retains large amounts of water within GAGs.

Proteoglycans
GAGs come in two different types. Proteoglycans are a single polypeptide that contains a few or many GAGs attached as side chains. Addition of GAGs to the protein occurs in the secretory pathway. The disaccharide side-chains are linked to proteins in the ER and Golgi. Proteoglycans include proteins of different lengths and different number sand types of covalently attached GAGs. Fibroblasts and other cells secrete proteoglycans through the secretory pathway.

Hyaluronan is another glycosaminoglycan but is unique because it is not attached to a proteins but exists as a free polysaccharide. Hyaluronan can contain up to 25000 repeats of a disaccharide and can reach a length of 20 µm, the size of an average cell. Hyaluronan lacks the structure of most proteins and contains many regions of that form random, flexible coils. Remember that the sugars in hyaluronan are negatively charged and repel each other. That generates a lot of space within the hyaluronan and allows it to occupy an incredibly large volume. In addition, hyaluronan like other GAGs bind and retains water which enables it to resist compression.

Digestions of Extracellular Matrix
Like many structures in the body, the components of the extracellular matrix must be replaced over time due to damage to the proteins that compose the ECM. Specific enzymes called matrix metalloproteinases (MMPs) digest the proteins in the ECM. MMPs can be secreted into the extracellular space or expressed as an integral membrane protein in the cell membrane. Fibroblasts synthesize new protein fibers to replace the digested components.

Digestion of the ECM also plays an important role in certain biological processes including development and immune responses.
Fibroblasts limit the extent of digestion of ECM by secreting proteins called tissue inhibitors of MMPs (TIMPs). TIMPs inhibit the activity to MMPs to ensure that the enzymes don’t degrade too much of the ECM which would impair the structure and function of cells in a tissue or organ. A balance between the amount of matrix metalloproteinases and inhibitors of matrix metalloproteinases is essential to maintain the integrity of the ECM while allowing for necessary biological process. Disease can arise when the balance between MMPs and TIMPs is significantly shifted.
